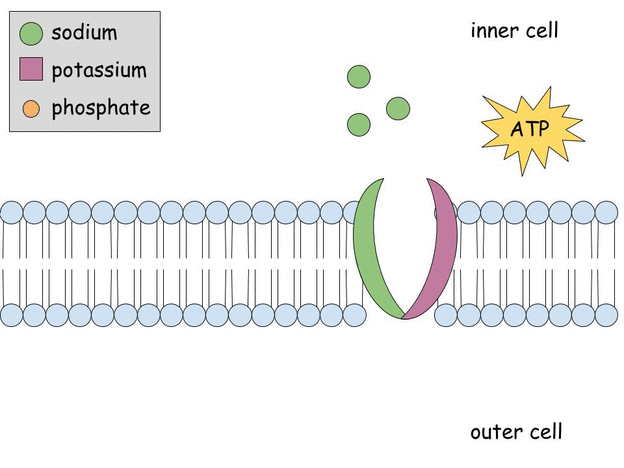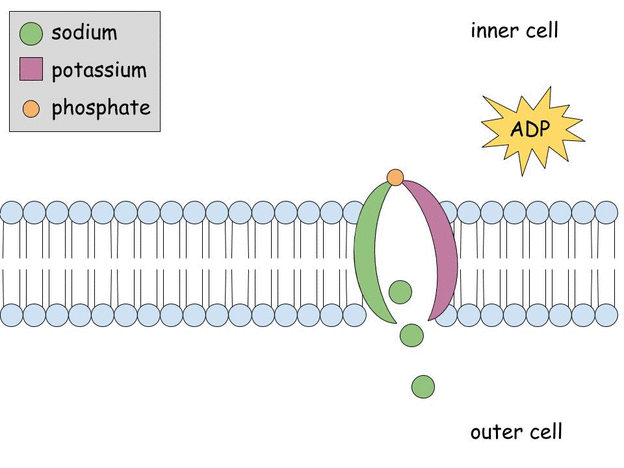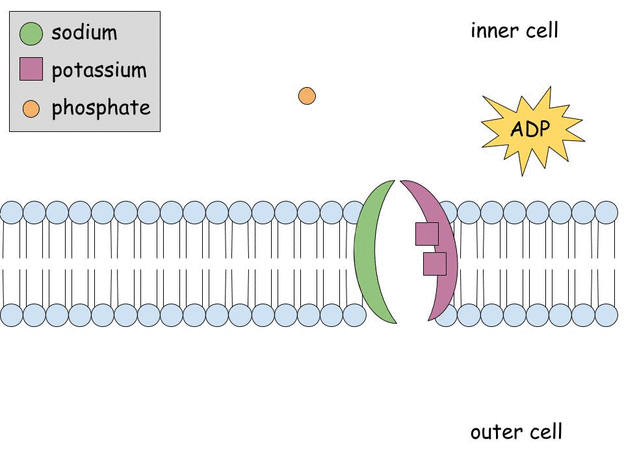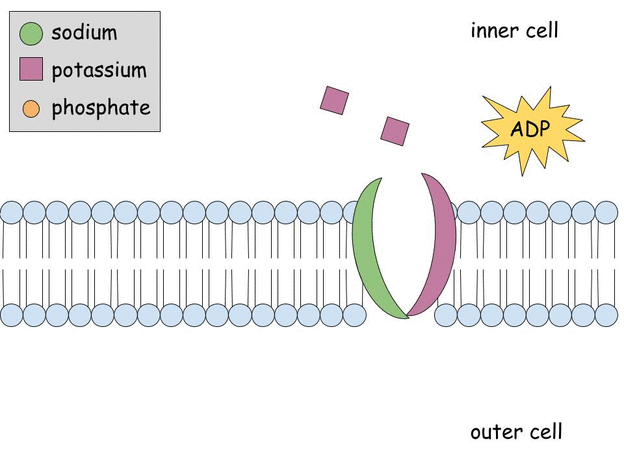
Aldosterone regulates Na+, K+ ATPase activity in human renal proximal tubule cells through mineralocorticoid receptor - ScienceDirect

Do H+ ions obscure electrogenic Na+ and K+ binding in the E1 state of the Na,K-ATPase? - ScienceDirect

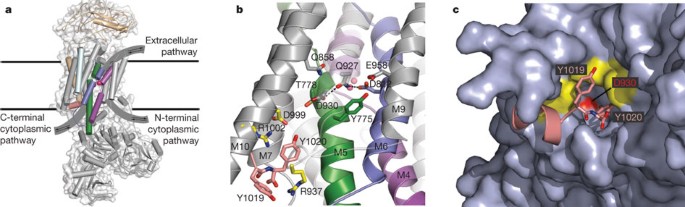
The selectivity of the Na+/K+-pump is controlled by binding site protonation and self-correcting occlusion | eLife
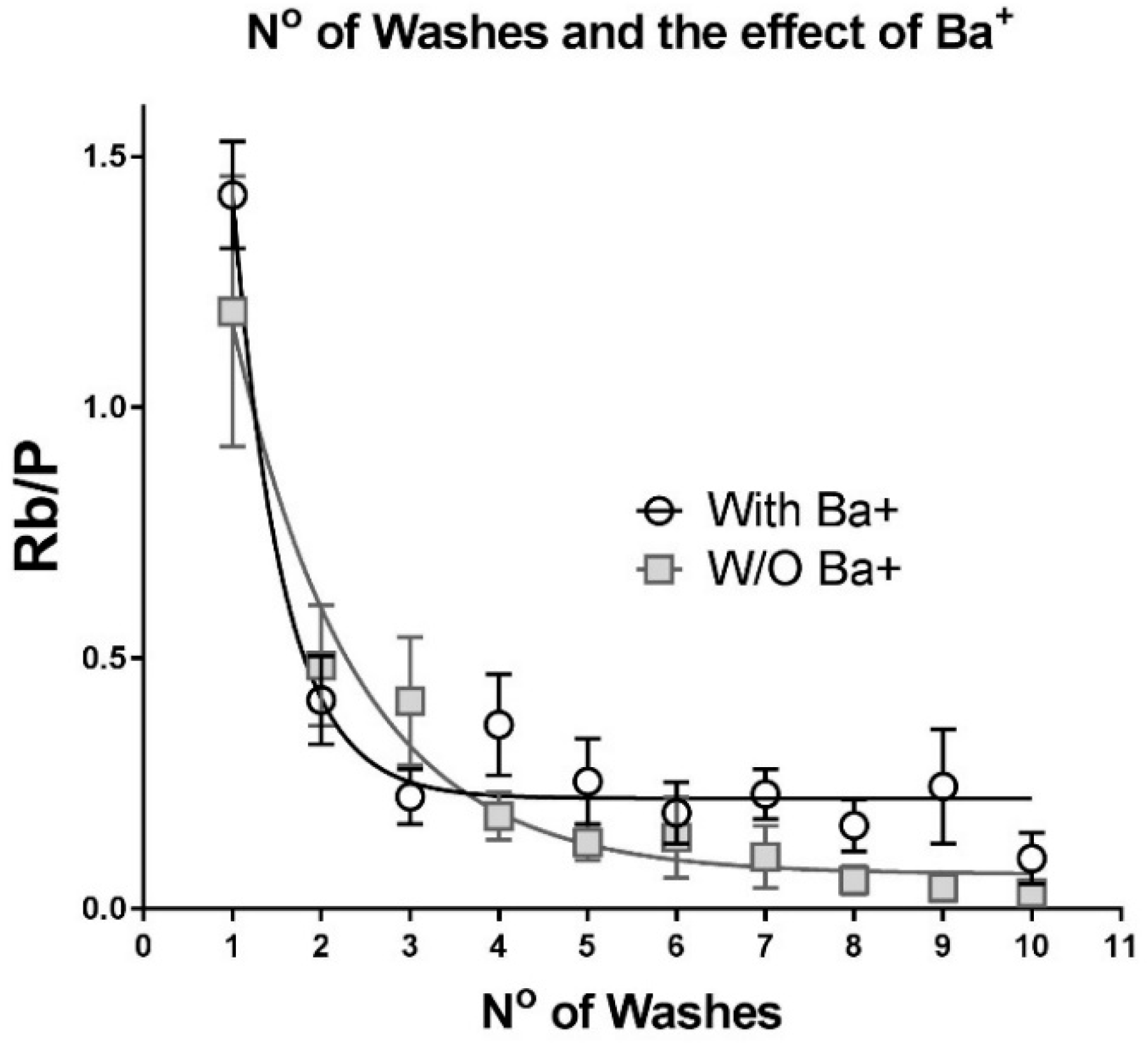
IJMS | Free Full-Text | K+ and Rb+ Affinities of the Na,K-ATPase α1 and α2 Isozymes: An Application of ICP-MS for Quantification of Na+ Pump Kinetics in Myofibers | HTML

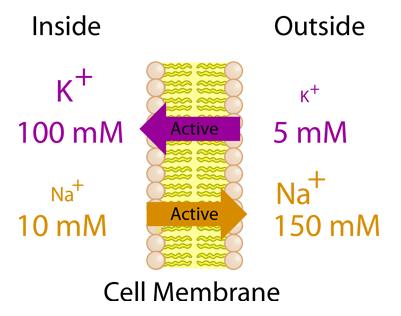
Human neuromuscular fatigue is associated with altered Na+-K+-ATPase activity following isometric exercise | Journal of Applied Physiology
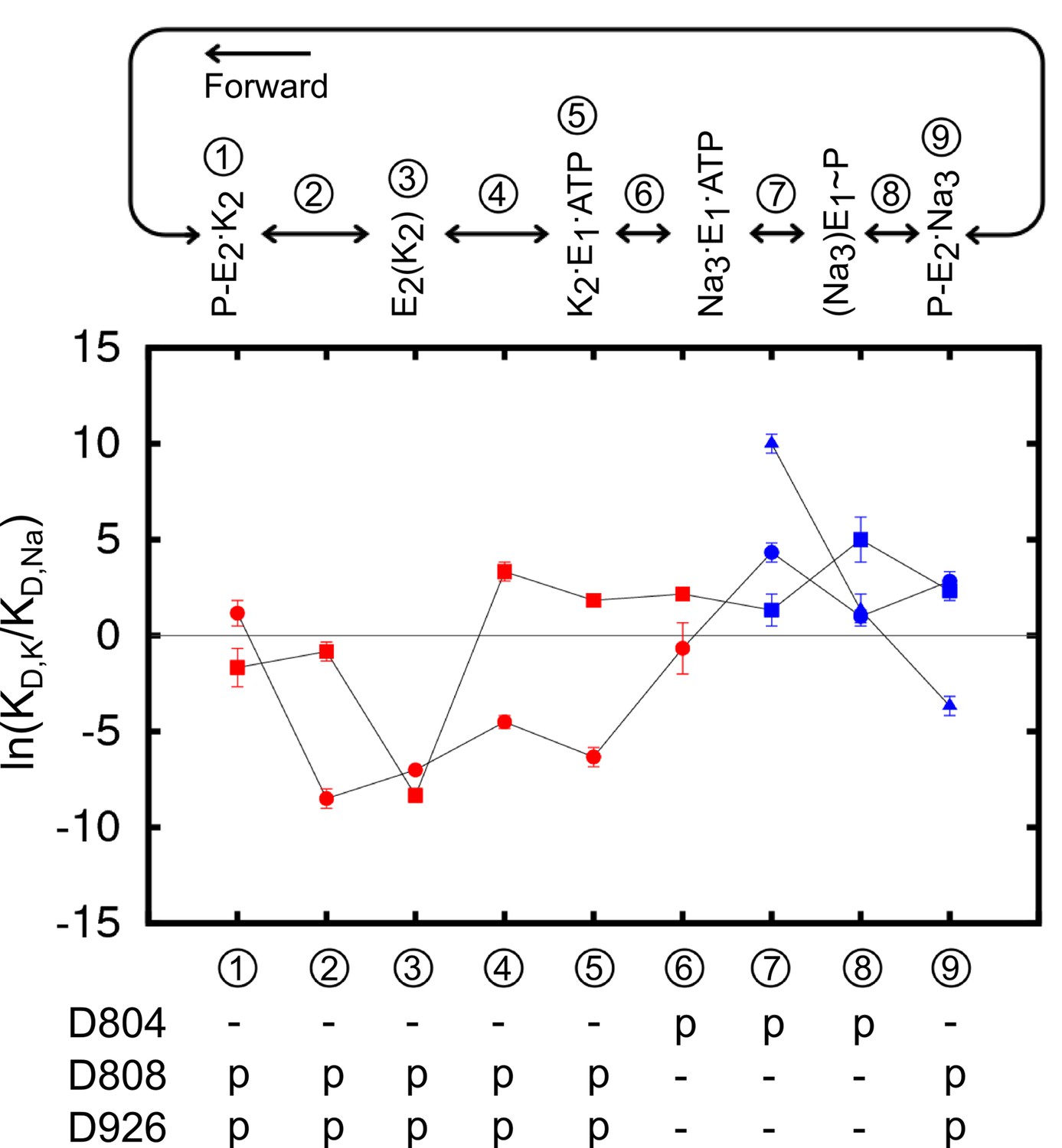
The selectivity of the Na+/K+-pump is controlled by binding site protonation and self-correcting occlusion | eLife

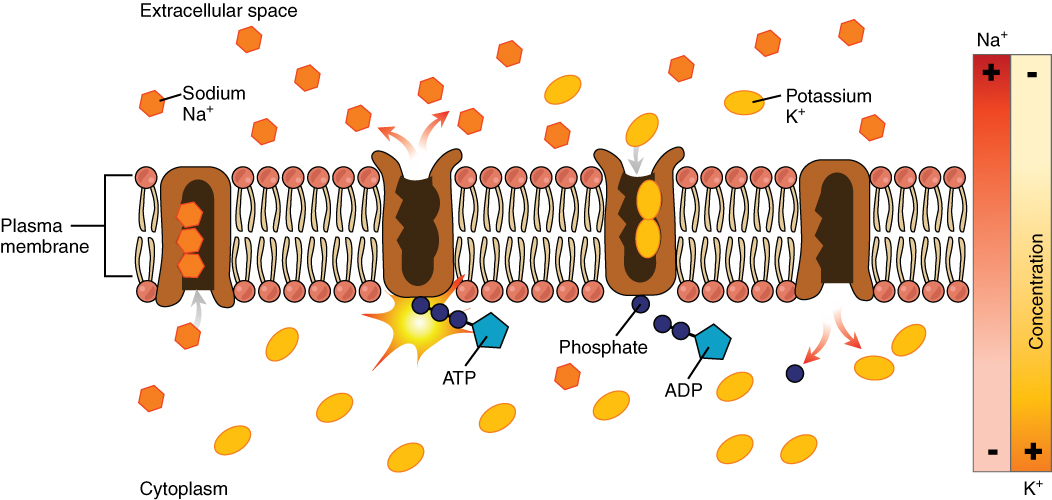
Kinetic contribution to extracellular Na+/K+ selectivity in the Na+/K+ pump - Vleeskens - 2018 - FEBS Open Bio - Wiley Online Library

Human neuromuscular fatigue is associated with altered Na+-K+-ATPase activity following isometric exercise | Journal of Applied Physiology
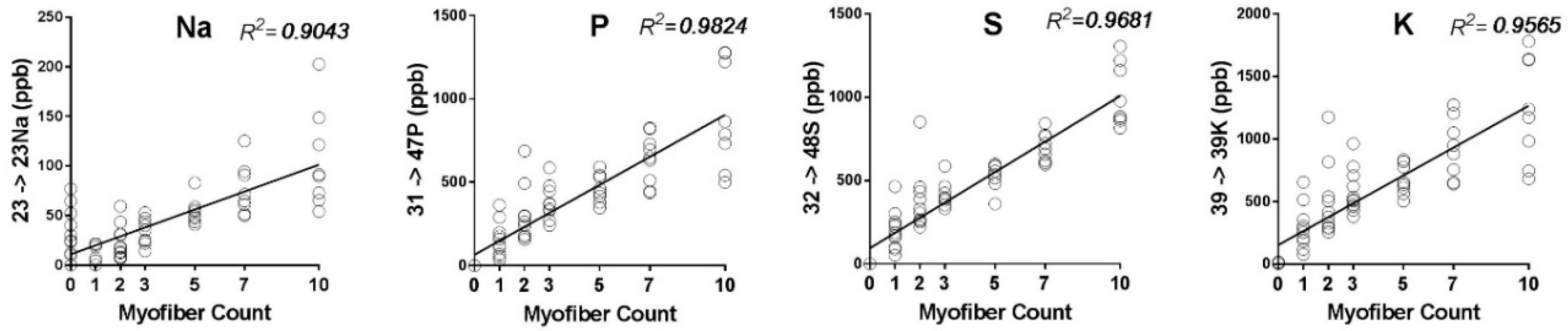
IJMS | Free Full-Text | K+ and Rb+ Affinities of the Na,K-ATPase α1 and α2 Isozymes: An Application of ICP-MS for Quantification of Na+ Pump Kinetics in Myofibers | HTML
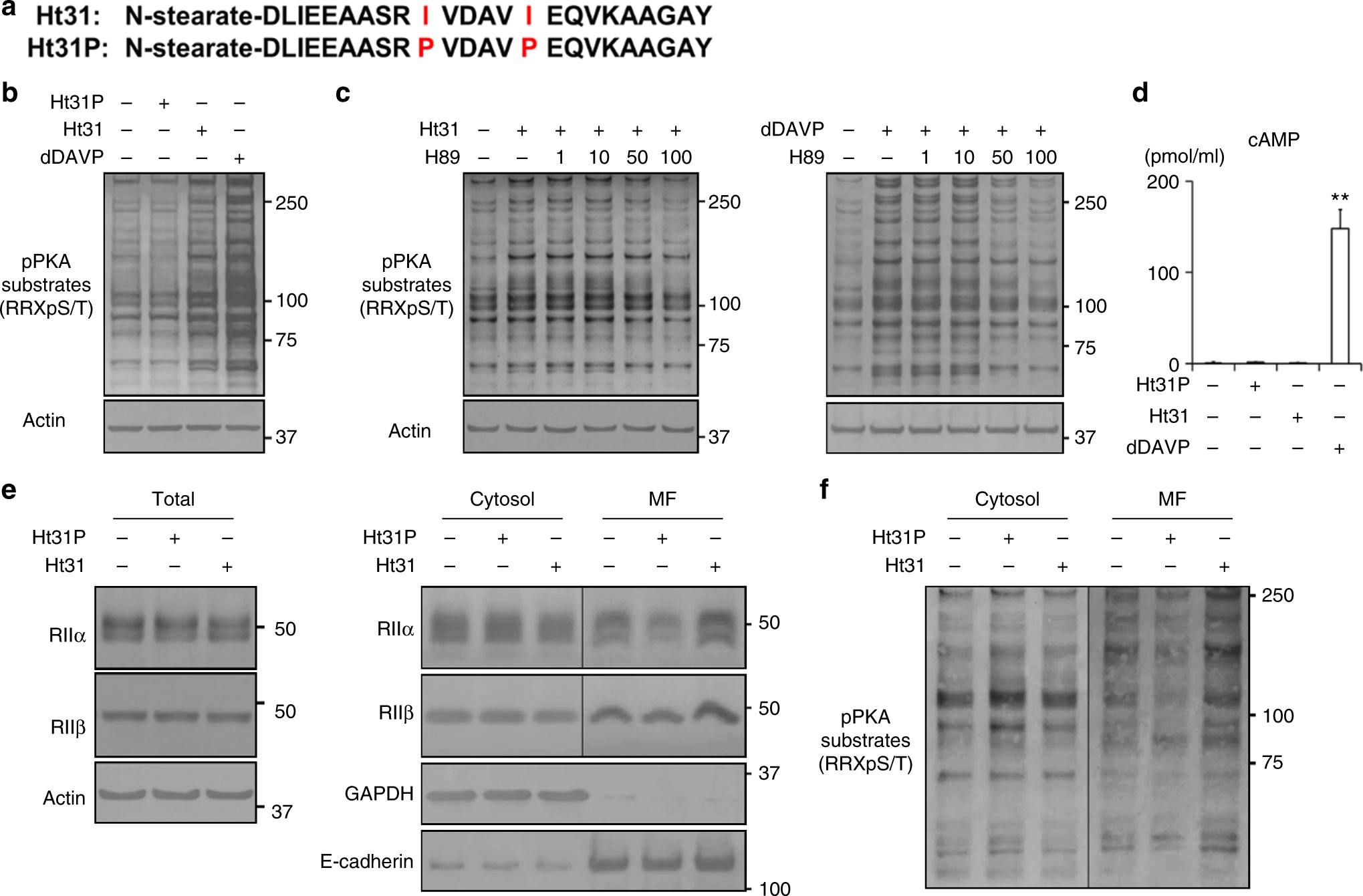
AKAPs-PKA disruptors increase AQP2 activity independently of vasopressin in a model of nephrogenic diabetes insipidus | Nature Communications

Aldosterone-induced protein kinase signalling and the control of electrolyte balance | Semantic Scholar

The selectivity of the Na+/K+-pump is controlled by binding site protonation and self-correcting occlusion | eLife
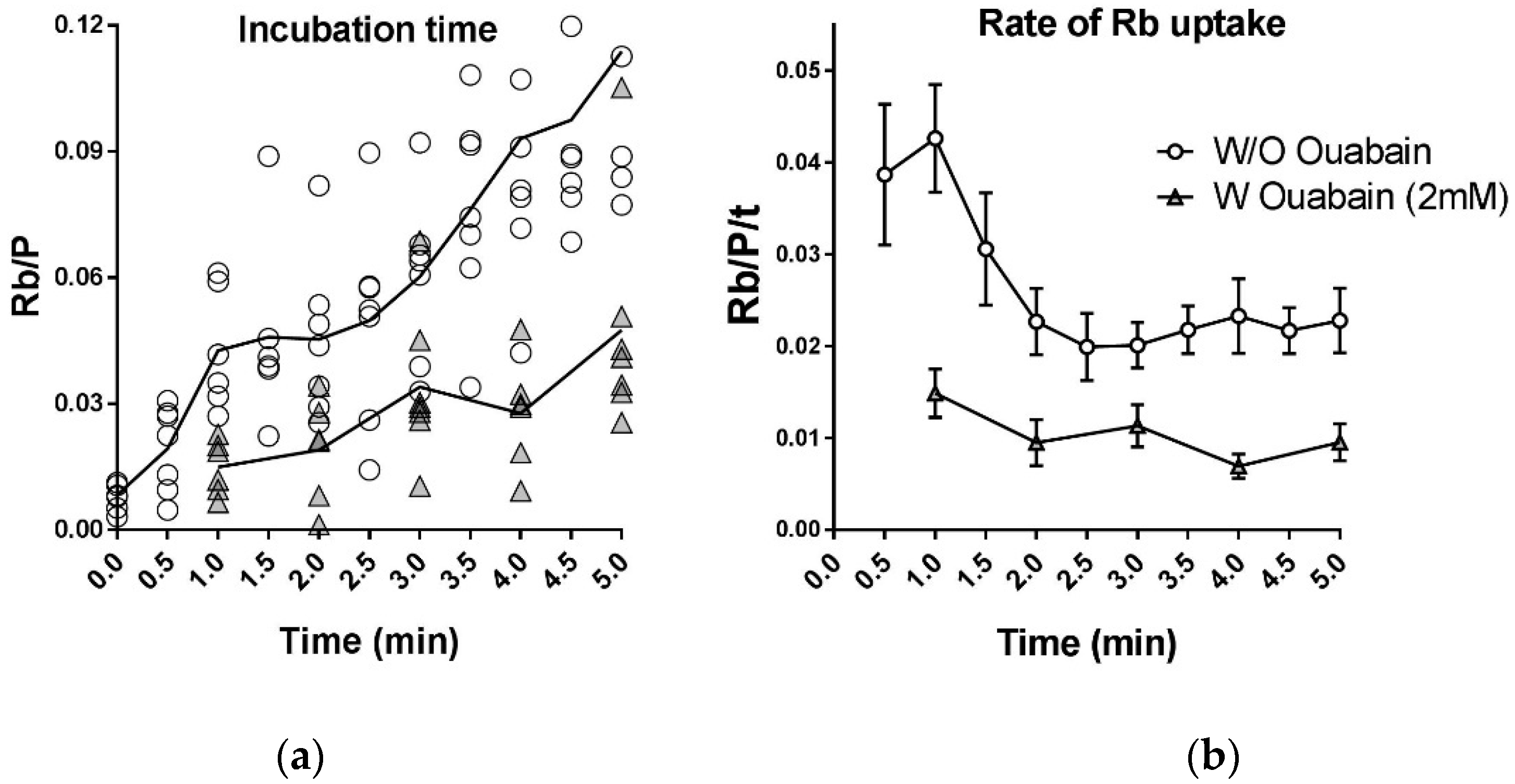
IJMS | Free Full-Text | K+ and Rb+ Affinities of the Na,K-ATPase α1 and α2 Isozymes: An Application of ICP-MS for Quantification of Na+ Pump Kinetics in Myofibers | HTML